CLEVELAND — Even as Ohio crossed a million total cases of the coronavirus Monday, many feel optimistic as AstraZeneca announced the results of their vaccine study, showing 79% efficacy in preventing symptoms of COVID-19, and complete protection against hospitalizations from the virus.
Dr. Abhijit Duggal serves as a staff ICU physician and director for critical care research for the medical ICU at the Cleveland Clinic.
“The most encouraging thing is it’s really effective in decreasing the risk of severe disease,” he said. “We want to have as many tools as we can and are available to use. That gives us the ability to protect more and more people.”
While the AstraZeneca two-shot vaccine has been authorized in more than 50 countries worldwide, it has not yet been approved in the U.S due to what Dr. Duggal calls a stricter approval process by the FDA and medical community.
“Before we approve anything in the U.S., we will do our due diligence,” he said. “The initial results are very promising and I think we need to look at this as another option available to us.”
This vaccine had faced opposition in Europe over alleged side effect concerns involving blood clots, with several countries temporarily stopping administration of the vaccine. Several days later, the EU's top medical agency said that the AstraZeneca vaccine was not the cause of that clotting issue, which has only occurred in a handful of patients out of the more than 17 million who received a dose.
Dr. Duggal told News 5 caution still needs to be exercised until all of the details of the study have been released by AstraZeneca.
Vaccine access continues to expand with Ohio announcing its plans to allow vaccine appointments later this month to everyone 16 and older, as well as allowing providers to open up unfilled appointment slots.
At the FEMA mass vaccination site at Cleveland State University’s Wolstein Center, more than 31,000 have received their first dose already, on pace for their planned goal of administering up to 6,000 people a day, Governor Mike DeWine said Monday.
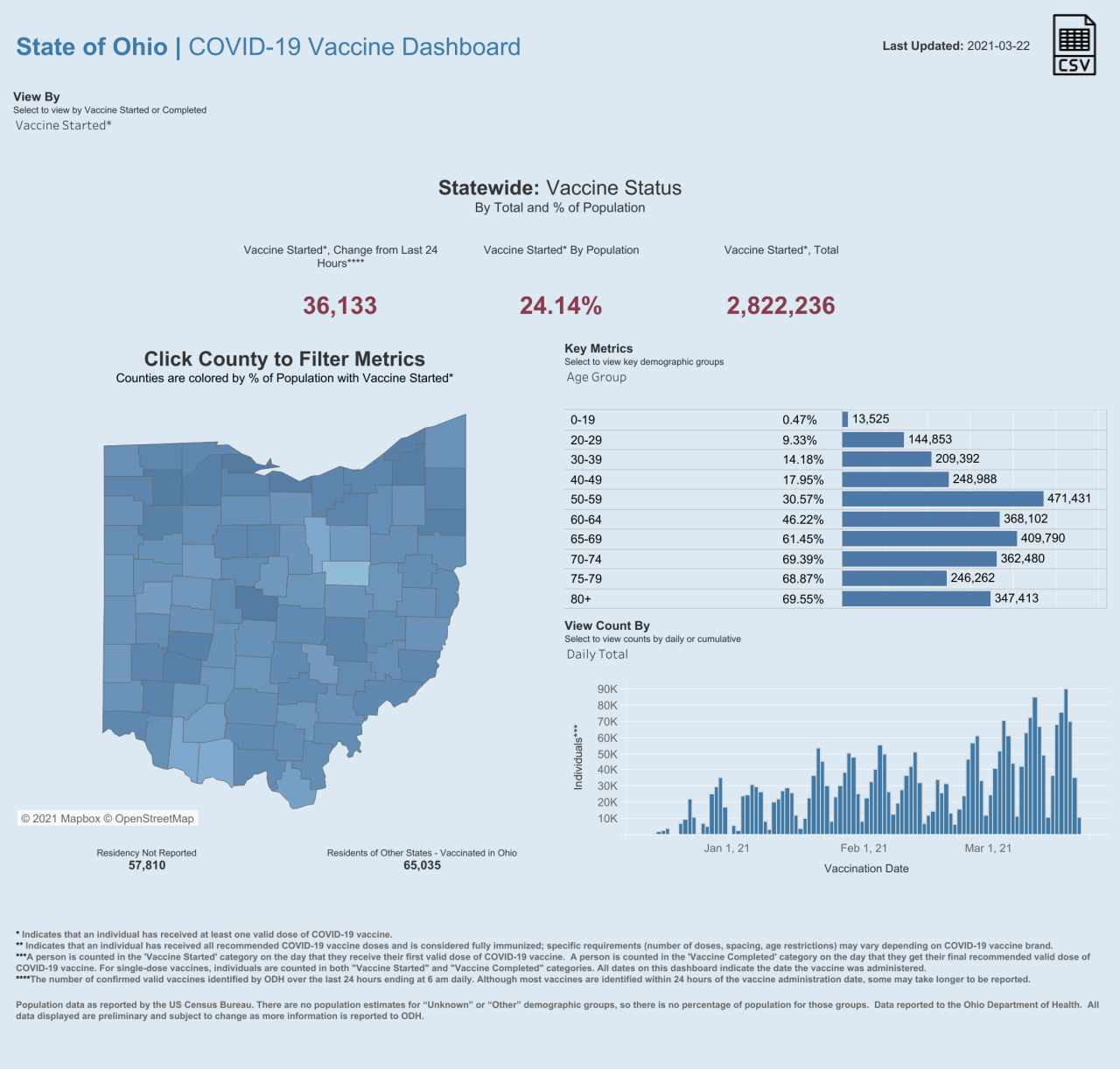
Statewide, the Ohio Department of Health website details how nearly a quarter of the state's population has received at least one dose of the vaccine.
AstraZeneca is expected to apply for emergency use authorization for its vaccine from the Food and Drug Administration vaccine sometime in early to mid-April.
Visit our Vaccinating Ohio page for the latest updates on Ohio's vaccination program, including links to sign up for a vaccine appointment, a map of nearby vaccination sites, a detailed breakdown of the state's current vaccine phase, and continuing local coverage of COVID-19 vaccines in Northeast Ohio.
Download the News 5 app for free for your Apple or Google device for the latest vaccine and COVID-19 news, and for limited and timely news alerts on vaccines, the COVID-19 pandemic, and other important local news and weather.





